The monkeypox virus is only one mutation away from evading a key antiviral drug being used to treat at-risk patients, federal health officials are now warning — and they’re urging doctors to be “judicious” in prescribing the sought-after treatment.
The new FDA guidance for the antiviral drug known as tecovirimat, or Tpoxx, was published this week online and in updated labeling.
The regulator says lab and animal studies, and evidence from a human case of this family of viruses, suggest monkeypox has “several genetic pathways” to evolve resistance to tecovirimat. Many “require only a single amino acid change,” the FDA said.
“Most patients with intact immune systems really need supportive care and pain control, but often do not need to be stepped up to antiviral treatment,” the Centers for DIsease Control and Prevention’s Dr. Sapna Bamrah Morris said over the weekend, in a webinar hosted by the Infectious Diseases Society of America.
The CDC has been closely tracking mutations in the virus to watch for potential variants that could be resistant to the monkeypox treatment. A portion of positive tests around the country are being forwarded to the agency for genetic sequencing.
One recent group of cases led to the CDC warning labs earlier this month of a “rare” mutation spotted in California that caused “false negative” results on some tests.
No cases have been spotted so far of a strain that can evade tecovirimat, though authorities warn this “low barrier to resistance” poses a risk that a resistant variant could emerge and spread.
“We do know that there’s a point mutation that could lead to resistance to tecovirimat or Tpoxx, which is our primary antiviral being used,” said Bamrah Morris.
The FDA’s warning comes as federal health authorities have been urging doctors to direct monkeypox patients to recently-launched clinical trials by the National Institutes of Health to study the drug, instead of prescribing it themselves, when possible.
Tecovirimat is still available for doctors to obtain for their patients through an “expanded access” protocol set up by the CDC. White House officials have touted their efforts to cut down on the paperwork in order to get the drug through this so-called “compassionate use” channel.
The CDC recently reported early promising results collected from patients who have taken the drug through this protocol. Symptoms improved in three days on average.
However, without a control group, the CDC cautioned against drawing conclusions about the tecovirimat’s effectiveness based on patients treated this way.
“Data from randomized controlled trials are critically needed to address knowledge gaps related to efficacy, safety pharmacokinetics in humans with monkeypox, and to monitor for development of resistance to tecovirimat,” the FDA’s Dr. Adam Sherwat told doctors at a recent webinar hosted by the American Medical Association.
Siga Technologies, which manufactures tecovirimat, has been actively supporting the federal trial with supplies to study its drug. Tecovirimat is currently only approved for smallpox, a related orthopoxvirus.
But the company’s chief scientific officer tells CBS News that the FDA’s “low barrier to resistance” statement “might be overstated.”
“Poxviruses are DNA viruses. They have the ability to edit their DNA, as they are replicating, and correct mistakes. As a result, the mutation rate in a poxvirus is a hundred to a thousand fold lower than you would see in an RNA virus like COVID or influenza,” said Siga’s Dennis Hruby.
Hruby cited examples of the drug appearing to work to help patients clear out the virus, even after 82 days in the case of one person who developed a resistant strain.
“Working with our colleagues overseas, how they’re prescribing the drug, they are not giving it to people who have mild cases. But if the cases progress to where the patient is in extreme pain and discomfort, and/or about to be hospitalized, then they get treated,” said Hruby.
Many monkeypox patients have reported excruciating rashes and lesions from their infections, and recovery can take weeks. The CDC now tallies 22,630 cases nationwide.
A handful of people have been hospitalized for complications from the virus.
The CDC reported Tuesday on two cases of previously healthy young men who were hospitalized after the virus spread to their brain and spinal cord.
Health officials in Los Angeles on Monday became the first to confirm a U.S. death due to the virus, in a person who was “severely immunocompromised.” Another death reported in Texas is still being investigated to determine what role the virus played.
The Biden administration has also moved to stock up on supplies of another smallpox antiviral. Manufactured by Chimerix, the drug known as brincidofovir or Tembexa could offer an alternative to doctors if tecovirimat is sidelined.
“We do expect it to be available soon. And CDC is currently developing an expanded access IND protocol to help facilitate the use of this product as a treatment for monkeypox as well,” the CDC’s Brett Petersen told doctors at the AMA webinar.
Source: CBS Minnesota
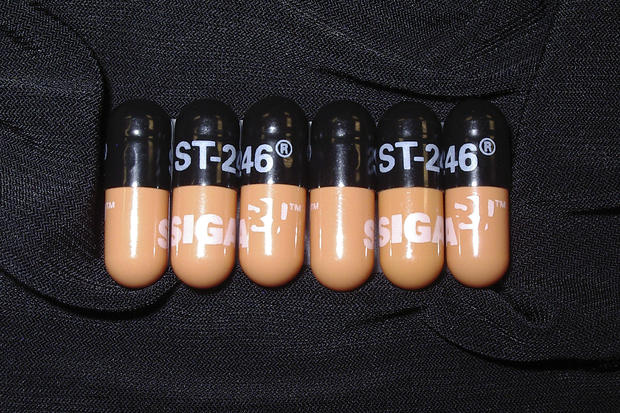
Be First to Comment